![]() 15 Minute Read May 14, 2026
15 Minute Read May 14, 2026
Labs and pathology groups are going digital faster than ever, and the ones moving now aren’t just chasing efficiency. They’re building the infrastructure for AI.
This guide walks through the decisions that actually matter when adopting digital pathology: what use case to start with, which scanner tier fits which setting, why slide management software is just as important as the hardware, and how to phase the transition without breaking daily operations.
Why 2026 Is a Different Conversation
Digital pathology has been “the future of pathology” for a long time. The difference now is that the barriers making adoption slow and expensive have largely come down.
The FDA cleared whole slide imaging for primary diagnosis in 2017. Scanner costs have dropped across every throughput tier. Software has matured to the point where going digital doesn’t require a six-month IT project to get started.
But the bigger shift is AI. Every AI-assisted diagnostic tool in pathology, whether it detects cancer, grades tumors, or quantifies biomarker expression, runs on whole slide images. A lab that hasn’t digitized its workflow can’t run any of those tools. Digital pathology adoption in 2026 isn’t just a workflow upgrade. For many institutions, it’s the first step toward AI readiness.

Choosing the Right Digital Pathology System: Where to Start
There’s no single right answer when it comes to choosing a digital pathology system. The right setup for a regional hospital running 80 slides a day looks very different from what works for a reference lab processing 500, or a teaching institution scanning slides primarily for education and research.
What makes the decision complicated isn’t the technology. Most serious vendors produce good images. What trips labs up is evaluating hardware before the underlying questions are answered: What problem is being solved first? How much volume needs to be handled? What does the workflow look like after the scan is done? And is the platform being chosen today going to support where the institution wants to be in five years?
This guide works through those questions in order. The goal is to arrive at a system that fits the actual workflow, not the most impressive spec sheet.

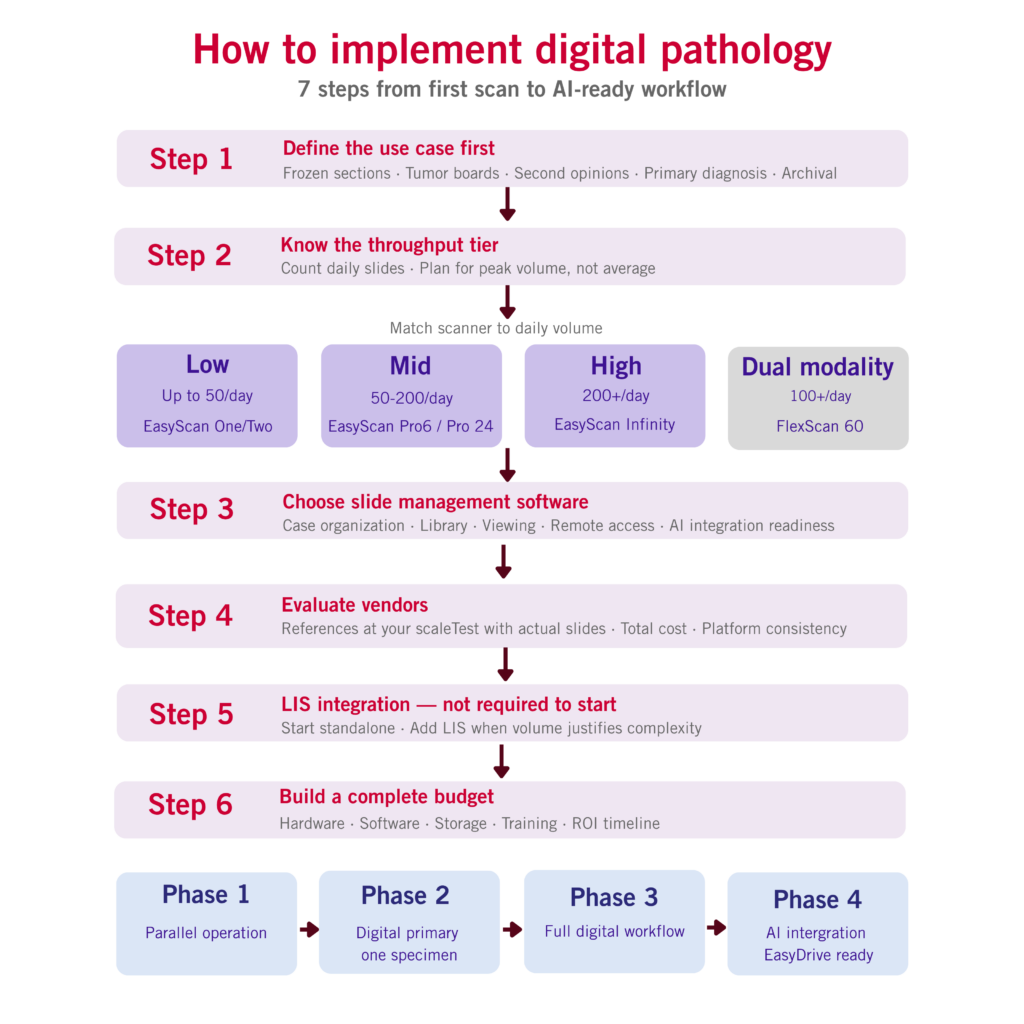
Step 1: Define the Use Case Before Looking at Hardware
The most common implementation mistake is opening with a scanner demo. The right starting point is a specific workflow problem that digital pathology will solve.
Vague goals like “we want to modernize” don’t translate into good procurement decisions. Specific use cases do.
Here are the most common starting points, roughly in order of simplicity:
Remote frozen sections. A pathologist reviews intraoperative tissue digitally from outside the surgical suite. No travel required, faster turnaround, and one of the clearest ROI cases in digital pathology because travel and courier costs are easy to calculate.
Tumor board and MDT presentations. Digital slides replace physical projection at multidisciplinary meetings. Zoom, annotate, and navigate in real time during case discussion. No formal validation is needed and value shows up from the first meeting.
Second opinion consultations. Whole slide images replace glass slides sent by post. No shipping delays, no breakage risk, and turnaround goes from days to hours.
Primary digital review for a defined specimen type. A pathologist signs out cases using digital images as the primary review medium. Formal CAP validation is required before going live, but this is the foundation of full digitization and the prerequisite for any AI workflow.
Archival scanning. Converting existing glass slide archives to digital. No formal validation is needed, but storage planning is critical.
Pick one use case, validate it properly, then build from there. Labs that try to go all-in on day one are the ones that stall.
Step 2: Know the Throughput Tier
Daily slide volume drives nearly every decision in digital pathology: scanner tier, storage requirements, software needs, and even implementation timeline. Count actual volume across all specimen types on a typical day, then plan for peak volume rather than the average.
Low Throughput: Up to ~50 Slides Per Day
The right fit for institutions beginning their digital pathology journey, satellite sites, and teaching labs validating one or two use cases before broader adoption.
The priority is simplicity. A compact scanner that deploys without major IT changes and doesn’t require specialized training to operate. It needs to perform on the institution’s actual tissue types, not just vendor demo slides.
Motic option: MoticEasyScan One / MoticEasyScan Two
Mid Throughput: ~50 to 200 Slides Per Day
The right fit for regional hospitals, multi-pathologist groups, and clinical settings where digital pathology needs to support daily diagnostic workflows rather than occasional use.
At this volume, multi-slide capacity, teleconsultation support, and reliable case management software all become essential. The image management platform starts to matter as much as the scanner itself.
Motic option: MoticEasyScan Pro 6 / MoticEasyScan Pro 24
High Throughput: 200+ Slides Per Day
The right fit for reference labs, academic medical centers, and hospital networks moving toward full digitization of primary diagnostic workflows.
Continuous, uninterrupted scanning. Automated quality control. Enterprise software with a LIS integration pathway. Vendor support also matters more here than at any other tier, because at this volume a scanner failure is a clinical event, not a minor inconvenience.
Motic option: MoticEasyScan Infinity
Dual Brightfield and Fluorescence: 100+ Per Day
Institutions running H&E or IHC alongside fluorescence imaging have historically needed two separate instruments, two maintenance contracts, and two scanning workflows. Dual-modality scanners consolidate both onto one platform, and the math on total cost of ownership often favors consolidation over five years.
Motic option: MoticFlexScan 60
Step 3: The Software Decision Labs Underestimate
There’s a pattern that shows up in many digital pathology implementations: a lab spends months comparing scanners and then picks software almost as an afterthought.
That’s the wrong order.
The digital slide management platform handles everything after a slide leaves the scanner: organizing cases, managing the image library, viewing and annotating slides, and sharing cases with remote pathologists or external consultants. It determines day-to-day usability and staff adoption more than the scanner does. A technically excellent scanner paired with slow, confusing software produces one outcome: pathologists who stop using it.
At a minimum, the platform needs to handle the following:
- Case organization and workflow triage: As slides come off the scanner, they need to be sorted, labeled, and routed to the right pathologist or department quickly. In digital pathology this step is often called triage, not in the clinical sense, but in the same spirit: getting the right case to the right person without delay. The less manual work required here, the better. A good platform handles this automatically based on scan time, barcode, or case type.
- Library and archive management: Slide volume compounds fast. Within months an institution can have thousands of cases. The platform needs to make finding a specific case, specimen type, or scan date straightforward, without hunting through folders.
- Viewing and annotation: The viewer is where pathologists spend their time. It needs to be fast at every magnification, browser-based so it works from any location without local software installation, and should support annotation, measurement, and side-by-side slide comparison for things like IHC versus H&E review.
- Remote access and case sharing: Pathologists working from other sites, consulting remotely, or reviewing cases from home need the same access they would have in the building. The platform should also allow controlled sharing with external consultants without requiring them to install anything.
But in 2026 there is a fifth requirement most institutions aren’t evaluating yet: AI integration readiness.
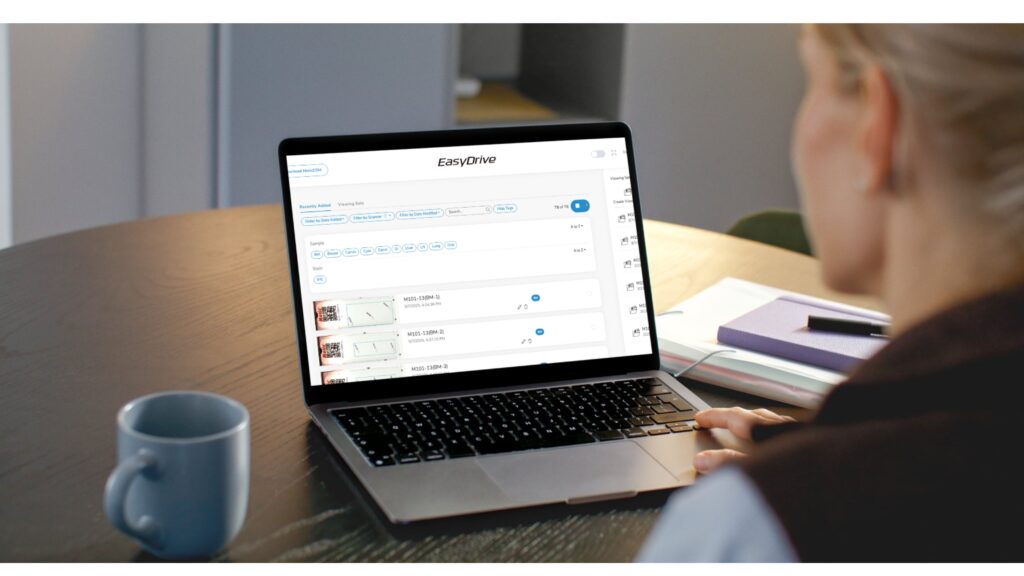
EasyDrive as a Gateway to AI
AI diagnostic algorithms don’t exist in isolation. Every tool that grades prostate cancer, quantifies HER2 expression, or flags regions of interest depends on two things: whole slide images and an image management platform to integrate through.
That platform is where AI outputs are surfaced to the pathologist—within the same viewer they already use.
A platform without an AI integration pathway creates a problem later. It forces a full software migration at the exact moment an institution is ready to adopt AI.
EasyDrive is Motic’s digital slide management platform, included with all Motic scanners. It supports the full workflow from scan to sign-out and is browser-based, so pathologists can access cases without local installation.
More importantly, the whole slide images managed today become the data layer AI tools will run on in the future.
Step 4: How to Evaluate Vendors
Image quality gets most of the attention in vendor evaluations. It matters—but it’s also where serious vendors are closest together.
The bigger differences show up elsewhere.
- Support coverage and response time: When a scanner fails before a frozen section, someone needs to be reachable immediately. Confirm whether support is genuinely available around the clock with staff located in the same region and get response time guarantees written into the service agreement.
- References from institutions at the same scale: Ask for references from settings with a similar caseload and use case, then call them directly. Ask about uptime over the past 12 months, how critical failures were handled, and whether they would choose the same vendor again.
- Testing with actual slides: Send the most challenging tissue types, thinnest sections, and any non-coverslipped specimens to the vendor before committing. A vendor who declines this request is telling you something important about how they handle problems after the sale.
- Total cost of ownership: The hardware price is the number vendors lead with. The full cost includes annual software licensing if not included, on-site service terms, and storage costs that scale over time. A scanner that looks cheaper at purchase can cost significantly more over five years.
- Platform consistency across the range: If the institution grows from a low-throughput scanner to a higher-throughput model, does the case archive carry forward? A vendor whose full range runs on the same platform protects the implementation investment as needs evolve.
Step 5: LIS Integration Is Not Required to Start
Many institutions delay adoption because they assume LIS integration has to come first. It doesn’t.
Image management software can run as a fully functional standalone platform from day one. Slides are accessioned via barcode at the scanner, cases are organized in the software, and pathologists access images through the viewer without any LIS connection required.
LIS integration makes sense at higher caseloads where manual case entry creates real overhead, or when bi-directional data flow between the pathology reporting system and the slide management platform is genuinely needed. For most institutions starting out or running a pilot, that ROI is years away.
Start without it and add it when volume justifies the complexity.
Step 6: What the Budget Actually Needs to Cover
- Software licensing: Some vendors include image management in the scanner price. Others charge substantial annual fees on top. Clarify this before comparing prices because it’s the most common apples-to-oranges problem in digital pathology procurement.
- Storage: Storage is often underestimated. A single whole slide image can range from 500MB to several gigabytes. At 50 slides per day, this becomes meaningful monthly growth. At 200 or more slides per day, storage becomes one of the primary ongoing operating costs. Define the storage policy, whether cloud, on-premise, or hybrid, before going live.
- Implementation time: Staff training, workflow redesign, and validation protocols all consume time from people already working at capacity. This doesn’t usually make it into the hardware budget but it absolutely should make it into the project plan.
- ROI timing: Institutions with teleconsultation or remote frozen section workflows often see positive return within 12 months because travel and courier savings are concrete. Those going for full primary digital diagnosis typically build the ROI case over three to five years. Model against actual costs, not industry averages.
Step 7: How to Phase the Transition
Phase 1 is parallel operation. Scan cases already being reviewed on glass without changing the primary review medium. Validate image quality on real slides, stress-test storage and network at real volume, and build confidence before any change becomes mandatory. Most institutions spend four to eight weeks here.
Phase 2 is digital primary for one specimen type. Pathologists switch to digital review for one formally validated specimen type while glass slide review continues for everything else. CAP validation happens here. Workflow problems that weren’t visible during parallel operation tend to surface in this phase, and it’s much easier to fix them inside a contained scope.
Phase 3 is full digital workflow. Primary digital diagnosis across all validated specimen types, with glass slides reserved for exceptions. Not every institution gets here on the same timeline, and a hybrid model is the right long-term setup for many clinical settings.
Phase 4 is AI integration. Institutions that have completed Phases 1 through 3 already have the digital infrastructure AI requires.
On pathologist adoption. Bringing pathologists into scanner evaluation and image quality review from the start, rather than presenting a finished system for mandatory use, makes a significant difference. Peer adoption driven by internal champions consistently outperforms top-down rollouts.
Frequently Asked Questions
What is digital pathology?
Digital pathology is the practice of scanning glass microscope slides into high-resolution digital images called whole slide images (WSI) that can be viewed, analyzed, shared, and stored electronically. It enables remote review, teleconsultation, AI-assisted analysis, and digital case management without physical slide handling or shipping.
What is digital slide management software and why does it matter?
Digital slide management software handles everything that happens to a whole slide image after it leaves the scanner: case organization, library management, viewing, annotation, and case sharing. Poor software produces poor adoption regardless of scanner quality.
Why does slide management software matter for AI readiness?
AI diagnostic tools in pathology process whole slide images and integrate through the image management platform. A platform built with open AI integration capability allows institutions to add AI tools as they become clinically validated without replacing existing software infrastructure. A platform with no integration pathway requires a full migration at the moment the institution is ready to act.
How many slides per day are needed to justify adoption?
There’s no universal threshold. An institution processing 15 slides per day with frequent remote frozen sections might recover hardware cost within 12 months through eliminated travel costs alone. Define the use case first, then model ROI against actual expenses.
Is CAP validation required before using digital pathology for primary diagnosis?
Yes. CAP guidelines require a formal validation study at the institution, on its slides, with the specific scanner and software configuration before WSI can be used for clinical primary diagnosis. The CAP WSI validation guideline was updated in 2022.
Is LIS integration required before going live?
No. Image management software operates as a standalone platform from day one. LIS integration is a future phase added when caseload and complexity make it worthwhile.
What storage capacity is needed?
Plan for roughly 1 to 3GB per slide. At 50 slides per day that’s 50 to 150GB of new data daily. At 200 slides per day, monthly storage growth reaches 6 to 18TB.
How long does implementation take?
A single use case on a compact scanner can be operational within days. A full implementation with LIS integration, multi-site access, and validated primary digital diagnosis typically takes three to six months.
Is digital pathology reimbursed?
New CPT add-on codes introduced in 2024 allow institutions to bill for digital pathology digitization procedures. Coverage varies by payer and region. Involve the billing team and review current CMS guidance as part of financial planning.
Where Motic Fits
Motic’s scanner range covers every throughput tier: MoticEasyScan One and Two for lower-volume settings, MoticEasyScan Pro 6 and Pro 24 for mid-volume clinical workflows, MoticEasyScan Infinity for high-volume continuous scanning, and MoticFlexScan 60 for institutions that need both brightfield and fluorescence imaging on one platform.
Every scanner runs on EasyDrive, Motic’s digital slide management platform included in the scanner price. EasyDrive handles the complete workflow from scan to sign-out and is built as the image management foundation institutions will use to integrate AI diagnostic tools as they enter clinical practice.
An institution that starts with a compact scanner and grows to enterprise-scale deployment stays on the same platform throughout. No software migration, no retraining, no rebuilding the case archive.