How Motic Uses Digital Pathology to Combat Cervical Cancer
© tmc_photos // Adobe Stock
Cervical cancer is considered a leading cause of cancer-related deaths among women worldwide. Let that sink in — a cancer that’s largely preventable by the HPV vaccine and, at the very least, detectable with an annual Pap smear test, is still prominent.
Why? Because the fight against cervical cancer isn’t just about the disease itself. Too often, cervical cancer patients reside in low- and middle-income countries, which retells a grim and unfortunately timeless tale: Women across the world still not having access to the healthcare services they need. So, what did Motic do?
Tell a tale of its own.
A tale of two doctors
In the early 2000s, Dr. Alfred Böcking, then Head of Cytopathology at the University Hospital of Dusseldorf and Dr. Branko Palcic, Head of Cancer Imaging at the BC Cancer Agency, created the Cellular DNA Ploidy Analysis System.
This novel pathology technique, instead of relying on traditional morphological interpretation of tissues or individual cells alone, also directly measures the amount of DNA content within a cell, and uses that to determine if a cell is potentially cancerous.
The technique was originally intended to be a diagnostic tool to support reading difficult cases; however, the researchers soon realized that DNA-ploidy analysis could be automated via artificial intelligence and applied en masse for early detection screening, since the quantification largely reduced the need for human interpretation. It was an extremely cost-effective approach, and had the potential both for commercial viability and to heavily impact the fight against cancer in poorer countries.
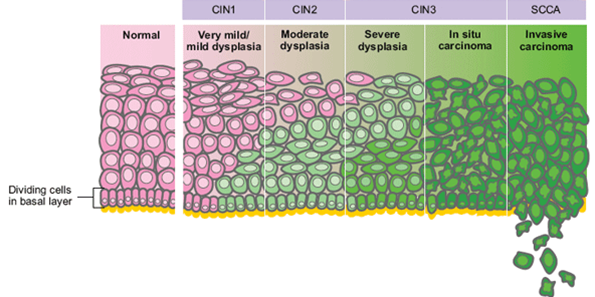
The development of cervical cancer // Source: TeachMe ObGyn
Still, the doctors needed help on the ground to promote and implement the nascent technology. Another colleague led Dr. Palcic to Motic and a partnership was formed to commercialize their technology in China. The timing couldn’t have been more opportune — Dr. Böcking and Dr. Palcic and the team at BC Cancer knew their technology could be used for widespread early detection of all types of cancer, and Motic knew just how much it was needed in China. While implementation would certainly be a pioneering endeavor, all parties involved were optimistic about the technology’s potential for success.
Combating cervical cancer in China
Relocated villagers in the Sichuan province // Xinhua
Motic has been a major player in enabling diagnosis and research in China since the 1980s, from microscopy tools to, more recently, digital solutions. Thus, the team was acutely aware of the gap in cancer detection and prevention services for the Chinese populace. Early detection screenings have a well-studied impact on reducing cancer rates in Western countries. However, 2010 data showed that only 21% of the female population in China reported having had regular screenings of any type. Much of China’s population also lives in rural communities. Coupled with a then major shortage of healthcare providers, finding a cost-effective option for widespread Pap smear screening felt impossible.
Enter: Motic and Drs. Böcking and Palcic’s DNA-ploidy system.
Because the DNA-ploidy system relies so heavily on automation, it negates the need for pathologists to make subjective interpretations of individual Pap smears. Technicians simply scan and upload the samples to an established cloud network using one of Motic’s WSI scanners, and then the DNA-ploidy software objectively analyzes each slide.
A Motic WSI scanner
There’s a clear advantage to this automated procedure: accessibility for a larger population. With the implementation of the system for widespread cervical cancer detection in China, patients are screened every two years, and a positive test result is quickly forwarded to a local specialist for treatment options. As a result of these routine cancer screenings, it’s much easier to detect cancer at an early stage, thus eliminating the need for intensive chemotherapy or surgery. Additionally, early stage cancer allows time for women in rural areas to travel to the city to receive proper treatment.
However, since DNA ploidy-conducted test results are still confirmed by human technicians, Motic realized an immediate need for standardization and quality control. The confirmation and general lab processes both require monitoring, so Motic built a cloud platform to maintain optimization. The platform intakes all results from all labs, every single day. Centralized monitoring is done by remote pathologists through Motic’s cloud platform, and feedback is delivered back to local hospitals as needed.
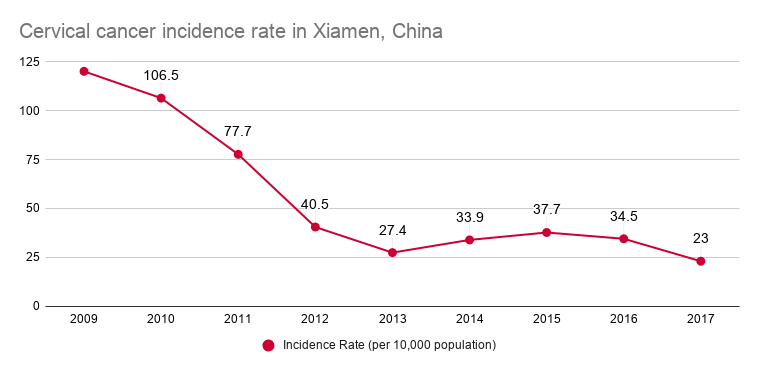
Cervical cancer incidence rate in Xiamen, China from 2009-2017, per 10K people // Source: Motic Medical
Through collaboration with local health authorities to implement digital pathology across China, Motic and its partners have helped dramatically decrease the cervical cancer incidence rate. In the city of Xiamen in particular, where Motic’s Chinese operations are headquartered, cancer incidence has decreased by 80% from 2009 to 2017, to 23.0 cases per 10,000 people.
Digital pathology as a long-term solution
Not only have Motic’s strong partnerships led to high-quality cancer diagnoses for millions of Chinese females across the country, but its telepathology platform for quality control can also be coupled with diagnostics services for even broader applications centered around closing the pathology care gap.
In western countries lacking schools for cytotechnicians, for example, Motic’s technology provides much needed support for a diminishing supply of qualified personnel who are trained to subjectively interpret Pap smears. Pairing digital pathology with artificial intelligence offers strong potential for scaling these services at a realistic pace.
In 2019, the World Health Organization announced an ambitious 10-year global strategy for combating cervical cancer, but there’s still plenty that can be done right now. With a combination of modern diagnostics technology and expert pathologist experience, the global health risk of cervical cancer can be mitigated.
If you’re interested in implementing digital pathology solutions for your lab, send us a message!